Seizure occurred in 0.6% of patients receiving XTANDI in eight randomized clinical trials. In a study of patients with predisposing factors for seizure, 2.2% of XTANDI-treated patients experienced a seizure. It is unknown whether anti-epileptic medications will prevent seizures with XTANDI. Patients in the study had one or more of the following predisposing factors: use of medications that may lower the seizure threshold, history of traumatic brain or head injury, history of cerebrovascular accident or transient ischemic attack, and Alzheimer’s disease, meningioma, or leptomeningeal disease from prostate cancer, unexplained loss of consciousness within the last 12 months, history of seizure, presence of a space occupying lesion of the brain, history of arteriovenous malformation, or history of brain infection. Advise patients of the risk of developing a seizure while taking XTANDI and of engaging in any activity where sudden loss of consciousness could cause serious harm to themselves or others. Permanently discontinue XTANDI in patients who develop a seizure during treatment.
Posterior Reversible Encephalopathy Syndrome (PRES) There have been reports of PRES in patients receiving XTANDI. PRES is a neurological disorder that can present with rapidly evolving symptoms including seizure, headache, lethargy, confusion, blindness, and other visual and neurological disturbances, with or without associated hypertension. A diagnosis of PRES requires confirmation by brain imaging, preferably MRI. Discontinue XTANDI in patients who develop PRES.
Hypersensitivity reactions, including edema of the face (0.5%), tongue (0.1%), or lip (0.1%) have been observed with XTANDI in eight randomized clinical trials. Pharyngeal edema has been reported in post-marketing cases. Advise patients who experience any symptoms of hypersensitivity to temporarily discontinue XTANDI and promptly seek medical care. Permanently discontinue XTANDI for serious hypersensitivity reactions.
Ischemic Heart Disease In the combined data of five randomized, placebo-controlled clinical studies, ischemic heart disease occurred more commonly in patients on the XTANDI arm compared to patients on the placebo arm (3.5% vs 2%). Grade 3-4 ischemic events occurred in 1.8% of patients on XTANDI versus 1.1% on placebo. Ischemic events led to death in 0.4% of patients on XTANDI compared to 0.1% on placebo. Monitor for signs and symptoms of ischemic heart disease. Optimize management of cardiovascular risk factors, such as hypertension, diabetes, or dyslipidemia. Discontinue XTANDI for Grade 3-4 ischemic heart disease.
Falls and Fractures occurred in patients receiving XTANDI. Evaluate patients for fracture and fall risk. Monitor and manage patients at risk for fractures according to established treatment guidelines and consider use of bone-targeted agents. In the combined data of five randomized, placebo-controlled clinical studies, falls occurred in 12% of patients treated with XTANDI compared to 6% of patients treated with placebo. Fractures occurred in 13% of patients treated with XTANDI and in 6% of patients treated with placebo.
Embryo-Fetal Toxicity The safety and efficacy of XTANDI have not been established in females. XTANDI can cause fetal harm and loss of pregnancy when administered to a pregnant female. Advise males with female partners of reproductive potential to use effective contraception during treatment with XTANDI and for 3 months after the last dose of XTANDI.
In the data from the five randomized placebo-controlled trials, the most common ARs (≥ 10%) that occurred more frequently (≥ 2% over placebo) in XTANDI-treated patients were musculoskeletal pain, fatigue, hot flush, constipation, decreased appetite, diarrhea, hypertension, hemorrhage, fall, fracture, and headache. In the bicalutamide-controlled study, the most common ARs (≥ 10%) reported in XTANDI-treated patients were asthenia/fatigue, back pain, musculoskeletal pain, hot flush, hypertension, nausea, constipation, diarrhea, upper respiratory tract infection, and weight loss.
In AFFIRM, the placebo-controlled study of metastatic CRPC (mCRPC) patients who previously received docetaxel, Grade 3 and higher ARs were reported among 47% of XTANDI-treated patients. Discontinuations due to ARs were reported for 16% of XTANDI-treated patients. In PREVAIL, the placebo-controlled study of chemotherapy-naive mCRPC patients, Grade 3-4 ARs were reported in 44% of XTANDI patients and 37% of placebo patients. Discontinuations due to ARs were reported for 6% of XTANDI-treated patients. In TERRAIN, the bicalutamide-controlled study of chemotherapy-naive mCRPC patients, Grade 3-4 ARs were reported in 39% of XTANDI patients and 38% of bicalutamide patients. Discontinuations with an AR as the primary reason were reported for 8% of XTANDI patients and 6% of bicalutamide patients.
In PROSPER, the placebo-controlled study of nonmetastatic CRPC (nmCRPC) patients, Grade 3 or higher ARs were reported in 31% of XTANDI patients and 23% of placebo patients. Discontinuations with an AR as the primary reason were reported for 9% of XTANDI patients and 6% of placebo patients.
In ARCHES, the placebo-controlled study of metastatic CSPC (mCSPC) patients, Grade 3 or higher ARs were reported in 24% of XTANDI-treated patients. Permanent discontinuation due to ARs as the primary reason was reported in 5% of XTANDI patients and 4% of placebo patients.
In EMBARK, the placebo-controlled study of nonmetastatic CSPC (nmCSPC) with high-risk biochemical recurrence (BCR) patients, Grade 3 or higher adverse reactions during the total duration of treatment were reported in 46% of patients treated with XTANDI plus leuprolide, 50% of patients receiving XTANDI as a single agent, and 43% of patients receiving placebo plus leuprolide. Permanent treatment discontinuation due to adverse reactions during the total duration of treatment as the primary reason was reported in 21% of patients treated with XTANDI plus leuprolide, 18% of patients receiving XTANDI as a single agent, and 10% of patients receiving placebo plus leuprolide.
Lab Abnormalities: Lab abnormalities that occurred in ≥ 5% of patients, and more frequently (> 2%) in the XTANDI arm compared to placebo in the pooled, randomized, placebo-controlled studies are hemoglobin decrease, neutrophil count decreased, white blood cell decreased, hyperglycemia, hypermagnesemia, hyponatremia, hyperphosphatemia, and hypercalcemia.
Hypertension: In the combined data from five randomized placebo-controlled clinical trials, hypertension was reported in 14.2% of XTANDI patients and 7.4% of placebo patients. Hypertension led to study discontinuation in < 1% of patients in each arm.
Effect of Other Drugs on XTANDI Avoid coadministration with strong CYP2C8 inhibitors. If coadministration cannot be avoided, reduce the dosage of XTANDI.
Avoid coadministration with strong CYP3A4 inducers. If coadministration cannot be avoided, increase the dosage of XTANDI.
Effect of XTANDI on Other Drugs Avoid coadministration with certain CYP3A4, CYP2C9, and CYP2C19 substrates for which minimal decrease in concentration may lead to therapeutic failure of the substrate. If coadministration cannot be avoided, increase the dosage of these substrates in accordance with their Prescribing Information. In cases where active metabolites are formed, there may be increased exposure to the active metabolites.
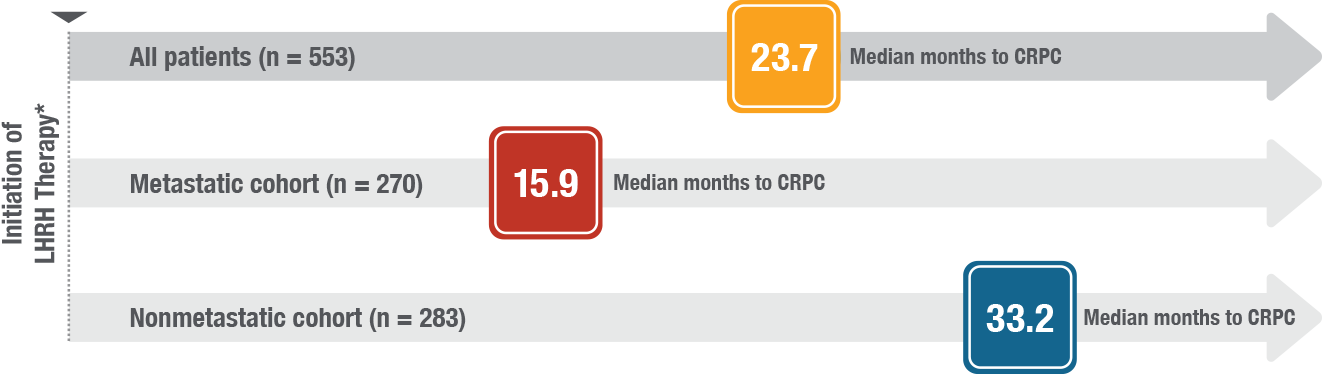
XTANDI® (enzalutamide) is indicated for the treatment of patients with:
- nonmetastatic castration-sensitive prostate cancer (nmCSPC) with biochemical recurrence at high risk for metastasis (high-risk BCR)
- metastatic castration-sensitive prostate cancer (mCSPC)
- castration-resistant prostate cancer (CRPC)
Please see Full Prescribing Information.